Check out my blog post about the process of organizing and executing WiSci's recent professional development symposium! http://wisciuga.weebly.com/blog/reflections-on-wiscis-first-professional-development-symposium
|
I recently gave a guest lecture (my first!) in Dr. John Maerz’s First-Year Odyssey Seminar, “Odyssey Into the Natural Sciences.” All freshmen are required to take one of 300+ FYO seminars, which are kept small (15-18 students) to promote student-faculty interaction. My prompt was to talk about emerging infectious diseases and the work I do with bats, in a 50 minute time slot. At first glance, this seemed like a fairly straightforward task, but I recognized some potential difficulties. Most of my experience in giving presentations is to ecology or disease-minded scientific audiences (MS/PhD holders and seekers). These talks are usually brief (10-20 minutes) and follow a typical outline: intro, methods, results, conclusions. For this talk, I had no idea what the scientific background of the students would be, so I didn’t want to get bogged down in data-heavy slides. Instead, I wanted to paint a picture of what it’s like to work in my system and some of the questions that we ask. Additionally, since this would be a longer talk, I needed ways to keep the students engaged as I spoke. Finally, I wanted to keep in mind the FYO goals (from the Faculty tab on the website):
My advisor gave me tips on how to structure my talk: start with a compelling hook, like the trailer for the 2011 movie Contagion, then broaden out to the larger picture of emerging infectious diseases (EIDs), how a lot of these have a zoonotic origin (i.e. from animals), and why we are seeing more in recent years. This would give us a common vocabulary to then explore the rest of my talk, about Hendra virus emergence and the work I’m doing as part of my PhD research. I wanted this section to be a little more personal, by sharing my first-hand experience of field and lab work in Australia. Using these tips, I developed a draft presentation that included lots of bat photos and a couple videos. I practiced the talk with my lab group, who gave me great feedback about where I could insert questions for the students to get them to think critically about the information I was providing (Goal 1: check) and maintain their attention over 50 minutes. They also pointed out areas where I was slipping into scientist mode, either by lapsing into jargon or not giving enough details to make a link clear. One lab member suggested including slides showing the departments we have at UGA working on wildlife, infectious disease, and public health (Goal 3: check). Giving my actual presentation to the class went well. The students responded when I asked questions, and based on eye contact and head-nodding, seemed to follow along with what I was saying. At the end, they had a lot of questions for me, especially about the Henda virus—flying fox system and working in Australia. Hopefully I’ll get the chance to do something like this again soon, to keep sharing my research and practicing my communication skills! At the ESA 100th Annual Meeting held recently in Baltimore, I gave an Ignite talk as part of a session on heterogeneity in wildlife disease ecology organized by Shweta Bansal. It was the first time I had given one of these talks, and I would guess that most other presenters hadn't given one before either. ESA introduced Ignite talks (although not the concept) at the 98th meeting (2013), so they're still relatively new. These talks have a strict format: 20 slides that auto-advance every 15 seconds, for a total of 5 minutes. In the email advice that was sent to presenters, ESA recommended that we watch other Ignite talks to get an idea of what to expect. I liked several of the suggested videos (especially the one on Fighting Dirty in Scrabble) but had trouble finding ones given by scientists. Googling "how to give a scientific Ignite talk" helped me find some general tips on preparation, but again, little in the way of tips from/for scientists. So here are my thoughts, two weeks now after my presentation, in the hopes that it might help future ESA or other conference presenters.
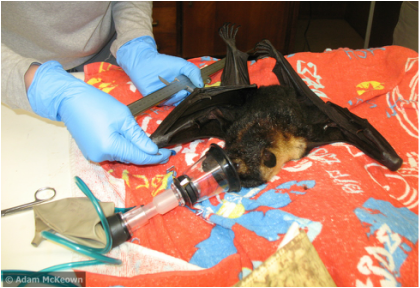
One more thing that I think would help future presenters: (if people are willing to share) ESA should start videotaping Ignite talks and posting them to the Ignite website. When preparing, I would have liked seeing how other scientists approached their talks. Maybe others have found successful ways to incorporate data (perhaps by "hacking" the format, e.g. repeating a slide to give yourself more time, although ESA told us not to). I recently had the chance to volunteer at the Tolga Bat Hospital, which was a great experience and a nice complement to my field work. Located in Atherton, Queensland, the bat hospital provides care for injured, ill, and orphaned megabats and microbats in the Far North QLD region (and sometimes further away). There is also a visitor center associated with the bat hospital which allows tourists and local citizens alike to learn about and observe bats. Jenny Maclean, with the help of volunteers, runs the facility and has done so for the past 25 years. This work requires an incredible commitment of time and energy; the bats are lucky to have such a champion as Jenny. During my week at the bat hospital, I helped with a variety of tasks. But since my experience is with flying foxes, I spent most of my time in their enclosure. There are about 100 adult flying foxes held in permanent care, often due to injuries that leave them unable to fly, or other conditions that would make it difficult for them to survive in the wild (i.e. blindness, cleft palate). Many activities revolved around feeding the bats--chopping and stringing up apples, hanging bird feeders packed with banana, filling small containers with banana smoothie and chunks of soft fruit, and hanging bottles of fruit juice supplemented with nutritional pudding. The bats subscribe to the philosophy of "easy come, easy go," so the remains of all of this food have to be cleaned up each morning. The copious amounts of spats (chewed, discarded mouthfuls of fruit that have had the juice sucked out), feces, and urine are scraped up and distributed into on-site worm farms. Then everything is hosed down, clean and ready for the bats to make a new mess. They get great room service! From 3-6pm, the visitor center is open for tours. A tour starts with a video introducing the work that goes on at the bat hospital, and then continues with a trip around to see all the bat enclosures (microbats, juvenile flying foxes, adult flying foxes, and the nursery). I was able to lead the flying fox portions of the tour. Normally I find public speaking to be stressful because I don't like when everyone's attention is focused on me. In this case, visitors were usually so engrossed by the bats that they only paid peripheral attention to me. Invariably during a tour at least one of the flying foxes would demonstrate their habit of flipping right side up (hanging with their thumb claws) to urinate/defecate, which always caused excitement. In my last post, I described how we capture flying foxes, but didn't get into what we actually do once we have caught them! So here is an update on that aspect of my work. The first step is putting the bat under anesthesia. Flying foxes are VERY squirmy, and it is safer for everyone to have them sedated while we are taking samples. We use inhalant isoflurane administered through a mask, which will usually put a bat to sleep in just a minute or two. The hardest part is actually finding the bat's head while it is still in the pillowcase, and placing the mask on top. Sometimes you will wonder why a bat is taking longer to go down than usual, and then you'll realize that you've been trying to anesthetize its rear end. Once the bat's legs have gone limp, we take it out of the pillowcase and start collecting data. The first things we measure are individual characteristics of the bat, like its species, sex, and age. We also measure its weight and forearm length as indicators of its condition. Then we start taking samples; for my PhD research, this includes fur, urine, and blood. Each of these samples can tell us different things about the bat it came from (topics for another post...). Once we have recorded all the information we need, we place the bat back into a clean pillowcase. We make sure that it has recovered from the anesthesia before releasing it back into the colony. Flying foxes cannot take off from the ground, so they need to be either released directly into the air or onto a tree. An air release means holding the bat as high up as you can with your gloved hand, otherwise it will try to climb up to the highest spot (i.e. your head). A tree release involves finding a rough tree trunk that the bat can grab onto with its claws. Bats are great climbers, and will quickly scramble up high before flapping away (likely a bit perplexed about what they have just experienced, but none the worse for wear).
I’m currently in Queensland, Australia conducting my first field season as a PhD student, so I thought I’d share one of my main activities while I’m here—catching the native flying foxes. Usually when I tell people I catch bats, I get a lot of confused looks, sometimes accompanied by a pantomime of waving a butterfly net back and forth in the air. Well, you’d need a pretty long net to catch these bats, which are flying at the height of tree canopies. Instead, we use something called a mist net—a black mesh net strung up between two poles (or tall trees, if there’s no room for poles) about 12-18 meters apart. The net is attached with a pulley on either side, and can be raised and lowered by two people, each standing at a pole. To use a mist net effectively, it has to be placed across the path of the flying foxes as they are coming back to the roost after a night of foraging. Flying foxes have good eyesight, but it is hard for them to spot the net on a dark night. When the moon is bright, you will see more bats flying over or around the net, although there always are some that still get trapped. We usually set up the net the day before we want to catch, in order to place all the ropes and clear any debris that might trip us. Bats usually fly back to the roost before dawn, so catching always involves getting up early—most recently 4 am on our last catching trip, but it can be even earlier depending on when sunrise is. Using our headlamps to guide us, we string the net across and raise it up, waiting for the bats to arrive. At the beginning, there aren’t too many bats around, but the pace picks up rapidly as the bats start to come back to the roost en masse. Everyone on the team (ideally, at least four of us) stares up at the sky, waiting for a bat to fly into the net. As soon as that happens, everyone turns on their headlamps and springs into action. The two people operating the net raise it down to around waist height. One handler, who has big gauntlets on, grabs the flying fox by the back of its neck so that it can’t bite anyone. This is harder than it seems, because often the neck is obstructed by netting. Once the head is secured, the other team members can go in and start untangling the bat’s claws and wings from the net. Flying foxes have very long, sharp claws, and can get tangled very quickly, so this step requires delicate fingerwork. Remarkably, most bats don’t squirm or make too much noise while this is going on. I’m not sure if they are just stunned by having big creatures point headlamps in their eyes, or understand that you are trying to untangle them, but it makes things easier. As pointy bits of the bat get freed, the handler tucks them under the big gloves so they can’t reach out and scratch anyone. Eventually, once the bat is completely free, the handler quickly releases it into a pillowcase held by another team member, which is subsequently tied closed and hung off a branch. Even a feisty bat will usually calm down at this point, as it can hang from the top of the bag in darkness and security. To be continued...
Back in October I started up Women in Science (WiSci) here at UGA with some other grad students in the Odum School of Ecology. Recently I submitted two applications on behalf of the group: one to the Parents and Families Association at UGA asking for a grant to support club activities, and another to UGA's Center for Student Organizations for the Outstanding New Organization Award. Although I submitted the applications as the group's president, many officers contributed to the writing--especially our fantastic and committed events coordinators. In reading over our final drafts, I was struck by how much we've accomplished as a group in less than 6 months.
Some highlights: 1. Membership recruitment: We started with a core group of six officers. Through outreach efforts like a press release, information sessions, a booth at the Winter Activities Fair, and campus-wide emails, we now have over 110 members from more than 25 departments: from Plant Sciences to Computer Science, from Forestry to Genetics, and more. 2. Mentoring program: Coordinated by our undergraduate liaison, our mentoring program matches undergraduate students with graduate student mentors to help mentees develop good study habits, choose majors, and apply for research opportunities and graduate school or jobs after graduation. We already have 15 undergraduates and 27 graduate students in the mentoring program. Next we'll be expanding to include a faculty-grad student component as well. 3. Discussion events: Our first monthly discussion, entitled “Why Do Women Leave the Sciences?” had a great turnout of nearly 25 students. Trappeze worked pretty well as a venue, and I definitely think that having the discussion off-campus made for a more open and casual environment for sharing. We already have our next discussion topic lined up. “Working together: a call for inclusion conservation” will address the work and experiences of Dr. Meg Lowman, who is an awesome canopy ecologist. We've also arranged the opportunity for WiSci members to meet with Dr. Lowman. 4. Building a social media presence: Due to the hard work of our webmaster, WiSci has also built an online network to facilitate communication and support among group members, start discussions on hot media topics regarding equality in the sciences, and share resources. Our organization website includes links to campus resources, outside organizations that provide assistance to women in science and STEM fields, and funding sources and fellowships. A calendar of a variety of events (e.g. lectures, workshops, social gatherings) that are relevant to our mission can be found on our website as well. Plus we're on Facebook and Twitter. Way to go, Wisci! There are more online articles than I could list that have inaccuracies about bats. Usually I just grimace and ignore them, but this one from the Boston Globe the other day got me riled up enough to actually write a response. The article "Ebola response shows flaws in US system" focuses mainly on human capabilities of dealing with the introduction of a novel pathogen to the US. It only briefly touches on the role of wildlife as the reservoir for many zoonotic diseases (those that can be transmitted from animals to humans). The sentence that got my attention/made me cringe was, "Ebola is thought to originate in fruit bats that bite primates." (my emphasis added). Here is the response I wrote back to the article's author, in the hopes that she would correct that sentence. No dice.
Dear Ms. Freyer, Thank you for your recent article in the Globe about the Ebola response in the US. I am a PhD student at the University of Georgia studying emerging diseases in fruit bats, and have followed the Ebola outbreak with interest. I had one comment to make regarding your statement that "Ebola is thought to originate in fruit bats that bite primates." It is true that there are several lines of evidence to suggest that fruit bats may be the natural reservoir for Ebola virus:
Fruit bats play an important role in the ecosystem by pollinating native trees (and insectivorous bats, like the ones we have in the US, provide pest control). Misleading statements about bats can cause people to fear them unnecessarily and potentially lead to mass bat culls. I hope that you will amend your article in light of the information I have provided, and remove the phrase "that bite primates." Sincerely, Cecilia Sánchez Ph.D. student Odum School of Ecology University of Georgia http://ceciliaasanchez.weebly.com/ |
CAS
PhD candidate at UGA. Bat lover + zoonotic disease enthusiast. Archives
January 2016
Categories |











 RSS Feed
RSS Feed
